The Sinclair Lab has provided evidence that epigenetic changes are a conserved cause of aging. The RCM Hypothesis posits that aging involves epigenetic dysregulation due to the redistribution of chromatin-modifying proteins to DNA damage sites, altering gene expression and cellular identity. Experiments with the ICE mouse model showed that inducing DNA breaks accelerates this process, while interventions to stabilize epigenetic states mitigate aging phenotypes, suggesting a mechanistic link between DNA repair and epigenetic maintenance.

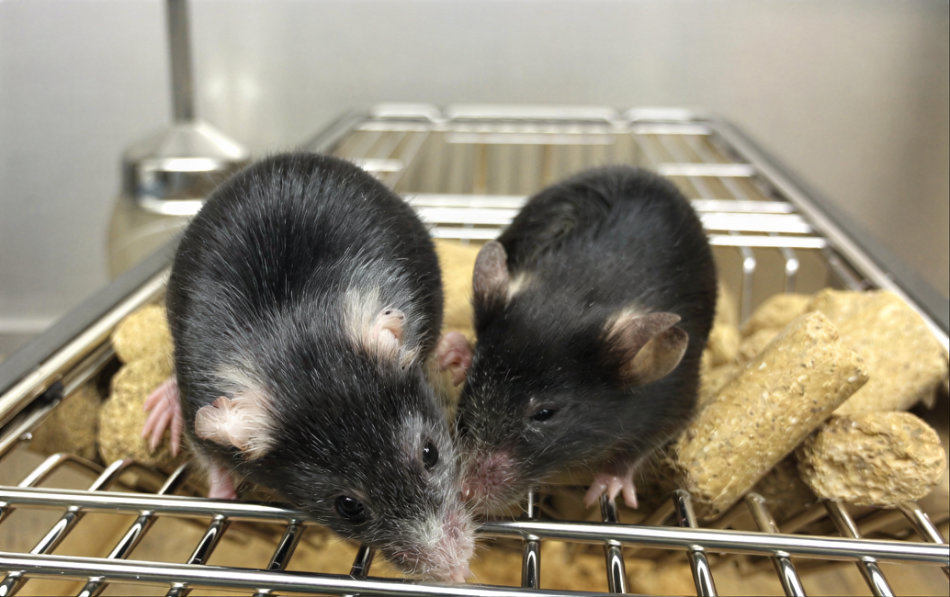
Legend: On the left, a normal 20 month old mouse and on the right, a sibling treated 16 months earlier with a system called ICE (for inducible changes to the epigenome) that was designed to test if epigenetic changes lead to aging in mammals
In a 2020 Nature study, the lab investigated epigenetic reprogramming in murine retinal ganglion cells. By expressing Oct4, Sox2, and Klf4, they observed a restoration of DNA methylation patterns and transcriptomes characteristic of younger cells, alongside enhanced axon regeneration and improved visual function in aged and glaucoma models. The findings indicate that mammalian cells retain latent epigenetic information that can be accessed to reverse aspects of cellular aging, providing a framework for studying age-related functional decline and potential therapeutic strategies (Lu et al., Reprogramming to recover youthful epigenetic information and restore vision, 2020)


Legend: In adults, the optic nerve does not regenerate when damaged. On the top panel, are orange-stained retinal ganglion nerves (RGCs) in the optic nerve after a nerve crush about 1/5 of the distance from the eye. On the bottom panel, is the same nerve type in a mouse treated with AAV-OSK, which expressed Oct4, Sox2 and Klf4 (OSK) in the RGCs for 6 weeks after the damage. Nerves are seen to survive the insult and regrow towards the brain, which was shown to be coincident with epigenetic age reversal and more youthful gene expression patterns that depended on DNA demethylation.
Dr. David Sinclair is a professor in the Genetics Department and an active member of the Paul F. Glenn Center for Biology of Aging Research, both at Harvard Medical School. He opened his lab 26 years ago, and his work in genetics and aging biology has fundamentally shaped the world’s understanding of mechanisms of aging and age-related diseases. His lab is best known for research on the molecular biology of aging, particularly the role of sirtuins, NAD+ metabolism, and epigenetic changes in regulating the aging process. His recent work provides evidence that cells retain a backup copy of youthful epigenetic information. Though its origin is unknown, this information can be accessed to reverse DNA methylation age and restore youthful gene expression patterns by expressing three transcription factors, Oct4, Sox2, and Klf4 (OSK) or treating cells with small molecule cocktails. Dr. Sinclair has been named in TIME’s Top 50 in Healthcare and 100 Most Influential People in the world. Additionally, other selected awards include an NIH Pioneer Award, Officer of the Order of Australia (AO), and Irving S. Wright Award of Distinction from the American Federation for Aging Research.

